Welcome to the IRB Office
Our mission is to protect the rights and welfare of human research participants. We are governed by 45 Code of Federal Regulations Part 46 (45 CFR 46), Protection of Human Subjects that dictate the scope and purpose of Institutional Review Board (IRB) activities, and follow ethical principles for the conduct of human research. The protection of research participants at Northwestern University is a shared responsibility, with the institution, researchers, IRB committees, and the IRB Office working together toward this common goal.
The Northwestern HRPP
The Northwestern University IRB Office directs the Northwestern University Human Participant Protection Program (HRPP) and is responsible for protecting the rights and welfare of human research participants. The HRPP's mission is to be a model program of excellence in protecting the rights and welfare of human participants involved in research.
- Further information about the Northwestern HRPP, qualifications of the Northwestern IRB Office, and research-related state laws is available here: Northwestern Local Context Information .
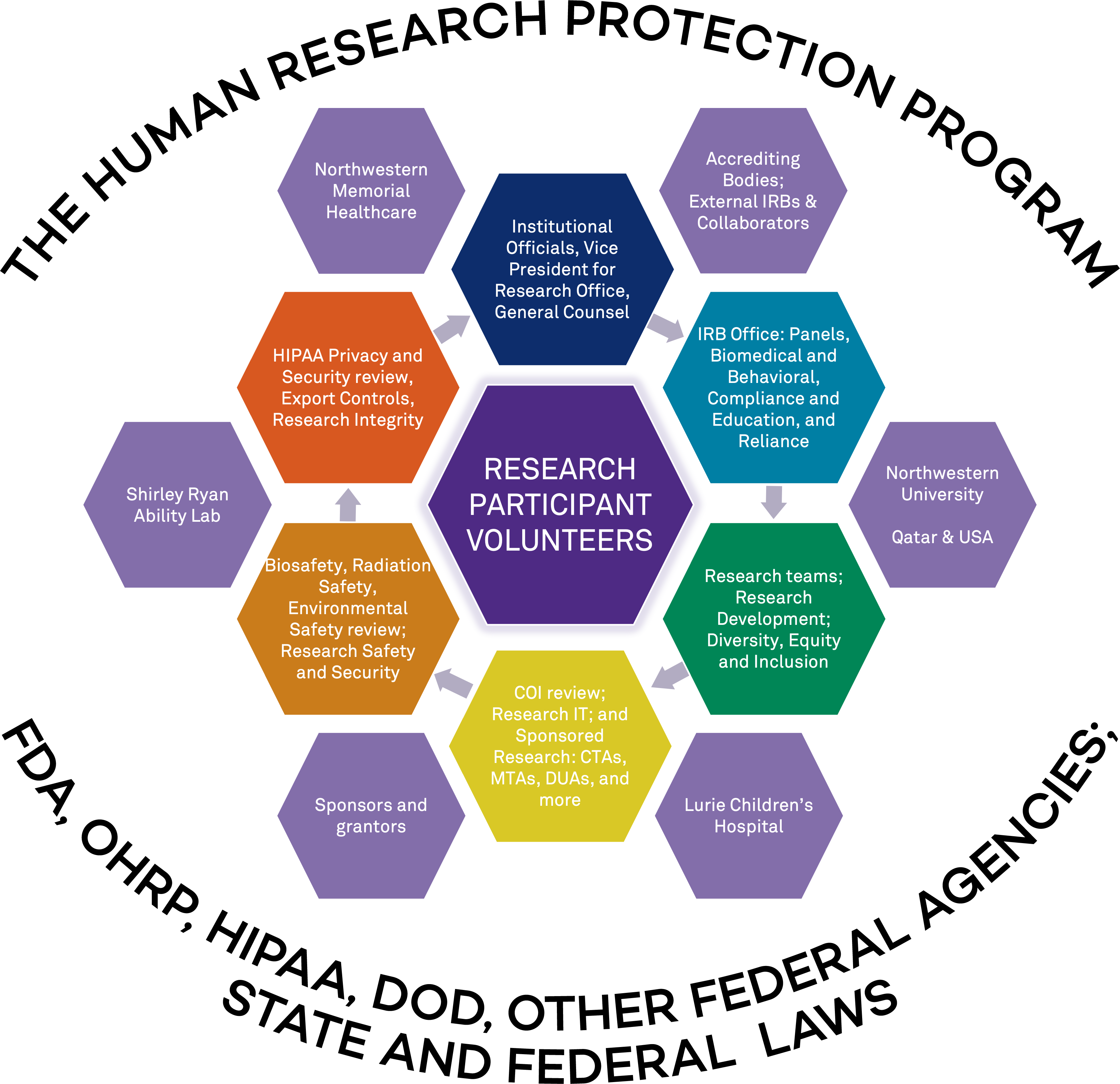
Entities Who Comprise Northwestern University's HRPP:
|
|
FWA and IRB Registration
Northwestern filed a Federalwide Assurance (FWA) with the Department of Health and Human Services (DHHS), in which we promise to uphold federal regulations and ethical principles for human subjects research conducted by employees, agents, and affiliates of NU.
- Northwestern University’s FWA number is 00001549
- Northwestern University's Federalwide Assurance (FWA) (Expiration: 12/12/2030)
The Food and Drug Administration (FDA) and Office for Human Research Protections (OHRP) require IRBs to register through a system maintained by DHHS.
- Northwestern University’s IRB Organization (IORG) number is IORG0000247
- Northwestern University’s IRB Registration Letter(Expiration: 12/17/2027)
Statements of Compliance
Good Clinical Practice (GCP) is an international ethical and scientific quality standard for the design, conduct, performance, monitoring, auditing, recording, analyses and reporting of clinical trials. GCP provides assurance that the data and reported results are credible and accurate, and that the rights, integrity and confidentiality of trial subjects are respected and protected. 21 CFR Part 11 is the FDA guideline that defines the criteria under which electronic records and electronic signatures are considered to be trustworthy, reliable and equivalent to that of the paper records.- View the NU Statement of 21CFR11 Compliance
- View the NMH Statement of 21CFR11 Compliance